
ATS 2023 International Annual Conference
Washington, DC
May 19-24, 2023
Learn how we are transforming the field of lung health at booth 2101
Stop by our booth to:
- Experience our trusted portfolio and latest technologies
- Test your skills with our fluoroscopic navigation bronchoscopy virtual reality experience
- See a patient management platform demo
- Ask questions and engage with our team of product experts
Seeing is Believing — Experience a virtual reality ILLUMISITE™ fluoroscopic navigation platform procedure to see the difference!
Join us in the booth to experience the ILLUMISITE™ platform with virtual reality!

Join our live events at ATS 2023

Guru Bar: ILLUMISITE™ platform as technology to aid in therapy delivery
Learn more about aiding therapy delivery with the ILLUMISITE™ platform, designed to correct for CT-to-body divergence — the discrepancy between a static CT scan and the breathing lung — with real-time visualization that enhances image clarity, and adjusts to a dynamic environment.1,2 Join Bryan Benn, M.D., PhD (Cleveland, Ohio) as he shares his experience during the ATS Guru Bar Presentations.
Sunday, May 21, 2023 – 1:30 p.m. – 1:50 p.m. ET
Featured at ATS 2023, booth 2101: Medtronic Lung Health Solutions innovative product portfolio
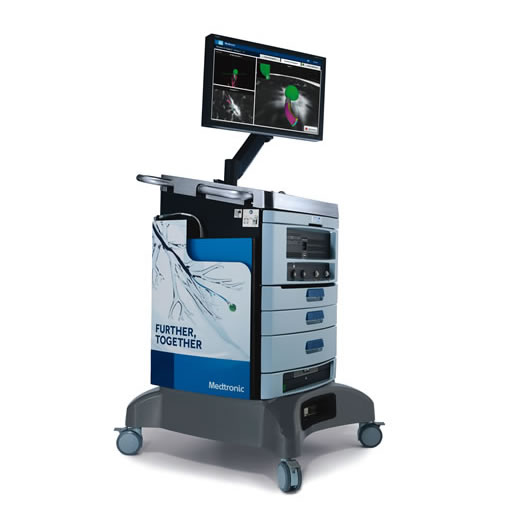
ILLUMISITE™ platform
Not only is the ILLUMISITE™ platform the leading electromagnetic lung navigation platform that corrects for CT-to-body divergence3,4 — it’s the first platform that used digital tomosynthesis to do so.1,3 The ILLUMISITE™ platform is clinically proven to help you accurately navigate lung nodules2-5,† and is designed for multidirectional sampling for a more thorough biopsy. And now it’s compatible with digital fluoroscopes, delivering expanded fluoroscopic capability during lung biopsy procedures.
LungGPS™ Patient Management Platform
Designed to assist clinicians in identifying incidental nodules and monitoring lung nodule patients, manage workflows, streamline thoracic tumor board collaboration, and provide analytics.
Meet the team
Get in touch with a Medtronic product expert or your local account manager to find the right solutions for you, your patients, and your facility.
† Based on evidence from a single-center prospective study including a total of 82 consecutive patients
1. Pritchett MA, Bhadra K, Mattingley JS. Electromagnetic Navigation Bronchoscopy With Tomosynthesis-based Visualization and Positional Correction: Three-dimensional Accuracy as Confirmed by Cone-Beam Computed Tomography. J BronchologyIntervPulmonol. 2021;28(1):10-20. doi:10.1097/LBR.0000000000000687
2. Avasarala SK, Roller L, KatsisJ, et al. Sight Unseen: Diagnostic Yield and Safety Outcomes of aNovel Multimodality Navigation Bronchoscopy Platform with Real-time Target Acquisition. Respiration. Published online September 03, 2021. doi:10.1159/000518009.
3. Pritchett MA, Bhadra K, Calcutt M, Folch E. Virtual or reality: divergence between preprocedural computed tomography scans and lung anatomy during guided bronchoscopy. J Thorac Dis 2020;12(4):1595–1611.
4. Medtronic financial and market analysis as of November 2021 of ENB and robotic lung navigation systems in market demonstrating Medtronic leading market share as defined by percentage of capital system sales.
5. Dunn BK, Blaj M, Stahl J, Speicher J, Anciano C, Hudson S, Kragel EA, Bowling MR. Evaluation of electromagnetic navigational bronchoscopy using tomosynthesis-assisted visualization, intraprocedural positional correction and continuous guidance for evaluation of peripheral pulmonary nodules. J Bronchology Interv Pulmonol. 2022.
The ILLUMISITE™ platform is indicated for displaying images of the tracheobronchial tree to aid the physician in guiding endoscopic tools or catheters in the pulmonary tract and to enable marker placement within soft lung tissue. It does not make a diagnosis and is not an endoscopic tool. Not for pediatric use.
WARNING: The ILLUMISITE™ platform may only be used by a qualified bronchoscopist.
CONTRAINDICATIONS: Flexible bronchoscopy should be performed only when the relative benefits outweigh the risks.
Absolute contraindications include, but are not limited to: lack of adequate facilities and personnel to care for emergencies such as cardiopulmonary arrest, pneumothorax, or bleeding, inability to adequately oxygenate the patient during the procedure.
Conditions that are usually considered absolute contraindications, unless risk-benefit assessment warrants the procedure: Coagulopathy or bleeding diathesis that cannot be corrected, severe obstructive airways disease, severe refractory hypoxemia, unstable hemodynamic status including dysrhythmias. Relative contraindications or conditions involving increased risk include but are not limited to: Recent myocardial infarction or unstable angina, uremia and pulmonary hypertension, and known or suspected pregnancy. Please refer to Instructions for Use for complete contraindication and risk information.
The LungGPS™ patient management platform is only available for sale in the United States.
The LungGPS™ patient management platform is an information management software only and does not diagnose or treat lung cancer nor replace lung cancer assessment in standard clinical practice. LungGPS™ should be used in conjunction with professional guidelines for patient management decision.
Contraindications: LungGPS™ is intended to integrate with specific file output of sequencing instruments such as Illumina sequencers as well as integrate with specific electronic medical records and external database warehouses. The system should not be used with any instruments that have not been tested and verified as compatible with LungGPS™.
©2023 Medtronic. Medtronic, Medtronic logo, and Engineering the extraordinary are trademarks of Medtronic. All other brands are trademarks of a Medtronic company. 03/2023–US-RE-2300187
Operational Headquarters 710 Medtronic Parkway, Minneapolis, MN 55432-5640 USA
©2026 Medtronic
